Biosurfactants are surfactants derived from biological organisms, and are rapidly growing in interest as a more sustainable alternative to petroleum and palm derived synthetic surfactants for product formulation. Like synthetic surfactants, biosurfactants are molecules that lower the surface tension between two materials, either a gas and a liquid (e.g. water surface tension), two liquids (e.g. water and oil), or a liquid and solid (water and dirt particles).
Biosurfactant Classification
There are many types of biosurfactants, which differ in their molecular structure and thus chemical properties. Biosurfactants can be broadly classified as low molecular weight (LMW) or high molecular weight (HMW). These can be further categorized based chemical structure:
- LMW biosurfactants include glycolipids, lipopeptides, and phospholipids
- HMW biosurfactants include lipoproteins, lipopolysaccharides, and complex biopolymers
Glycolipids, a class of LMW biosurfactants, are of particular interest due to their many commercial and industrial applications. These are characterized by a carbohydrate forming the hydrophilic “head” of the molecule, linked to a fatty acid forming the hydrophobic “tail”. Glycolipids are typically extracellular biosurfactants, meaning the microorganisms release the surfactants into the medium in which they are grown.
There is significant diversity in the molecular structure of the head and tail of glycolipids, resulting in the need for further subclassification. Subclasses of glycolipids include rhamnolipids, tehalolipids, and sophorolipids.
Sophorolipid Biosurfactants
Sophorolipids are a subset of glycolipids produced by several microbial species with a characteristic molecular structure. As implied by the name, sophorolipids are composed of a sophorose-based head and a fatty acid (lipid) tail. Sophorose is a glucose dimer – two glucose molecules bound together in a specific manner – and forms the hydrophilic carbohydrate head.
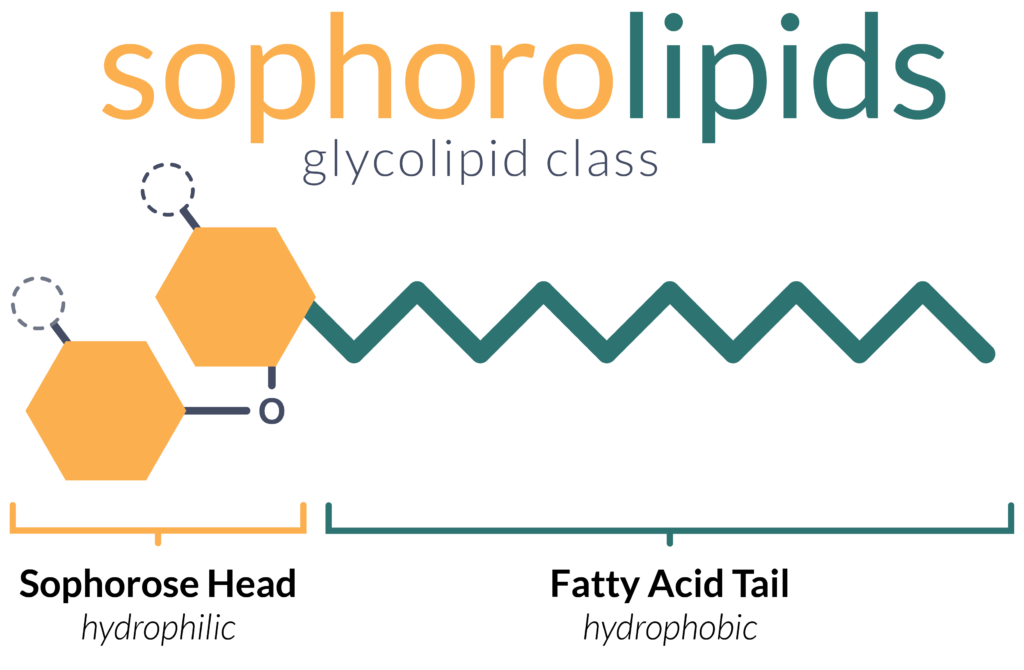
Sophorolipids are not a single molecule, rather, form a class of molecules in its own right. There are two main ways in which various sophorolipids differ:
- Modifications of the sophorose head: The most common modification to the sophorose head is acetylation – the attachment of acetyl-groups to the molecule, though other modifications are possible.
- Lactonization: The end of the fatty-acid chain can either be free (open/acidic form), or bound to the sophorose head to form a “loop” (lactonic form). Typically, lactonic sophorolipids are more efficient at lowering surface tension, while acidic sophorolipids exhibit better foaming properties.
Sophorolipids can further differ in other ways, including alternate or modified structures of the fatty acid chain. Different microbial species produce different types and mixtures of sophorolipids, which can be further engineered to change the desired structure of the biosurfactants produced. Chemical modifications can also be done following sophorolipid extraction to alter the molecular structure.
Properties & Applications
Differences in sophorolipid structure can significantly affect its properties, and it’s this variability that allows for a wide range of applications. Properties exhibited by various SLs include lowering surface tension, wetting, foaming, antimicrobial activity, degreasing, and emulsification.
Applications for sophorolipids can be found in a wide range of industries, including (but not limited to) in cleaning, cosmetics, pharmaceuticals, agriculture, and enhanced oil recovery (EOR). Due to the possible diversity in sophorolipid structure and characteristics, careful selection and design of biosurfactant formulations are essential to optimize for a desired application.
Sophorolipids are among the most promising class of biosurfactants for commercial use, with their increased adoption accelerating our transition to creating greener and more sustainable products.
References & Further Reading
- Costa JA, Treichel H, Santos LO, Martins VG. Solid-state fermentation for the production of biosurfactants and their applications. InCurrent developments in biotechnology and bioengineering 2018 Jan 1 (pp. 357-372). Elsevier. https://doi.org/10.1016/B978-0-444-63990-5.00016-5
- Kulakovskaya E, Kulakovskaya T. Extracellular glycolipids of yeasts: biodiversity, biochemistry, and prospects. Academic Press; 2013 Dec 11. https://doi.org/10.1016/B978-0-12-420069-2.00001-7
- Mnif I, Ellouz-Chaabouni S, Ghribi D. Glycolipid biosurfactants, main classes, functional properties and related potential applications in environmental biotechnology. Journal of Polymers and the Environment. 2018 May;26(5):2192-206. https://doi.org/10.1007/s10924-017-1076-4
- Roelants S, Solaiman DK, Ashby RD, Lodens S, Van Renterghem L, Soetaert W. Production and applications of sophorolipids. Biobased surfactants. 2019 Jan 1:65-119. https://doi.org/10.1016/B978-0-12-812705-6.00003-4